Seelos Therapeutics Announces Top Line Results from SLS-002 Phase II Study in Adults with Major Depressive Disorder at Imminent Risk of Suicide
- SLS-002 demonstrated early and persistent clinically meaningful reductions in symptoms of depression and acute suicidality
- Robust Response and Remission Rates were observed using the Montgomery-Åsberg Depression Rating Scale (MADRS)
- Results demonstrate the therapeutic potential of SLS-002 to address imminent suicidality
- SLS-002 was well-tolerated, with no evidence of new or unique adverse events and there were no deaths reported in the study
- The Company will host a webcast with Dr. David V Sheehan, today, Wednesday, September 20th at 8:00 a.m. ET
NEW YORK, Sept. 20, 2023 /PRNewswire/ -- Seelos Therapeutics, Inc. (Nasdaq: SEEL) ("Seelos"), a clinical-stage biopharmaceutical company focused on the development of therapies for central nervous system disorders and rare diseases, today announced top line data demonstrating clinically meaningful treatment effects across multiple endpoints and a well-tolerated safety profile from the double-blind, placebo-controlled cohort (Part 2) of its Phase II study of SLS-002 (intranasal racemic ketamine) for Acute Suicidal Ideation and Behavior (ASIB) in adults with Major Depressive Disorder (MDD).
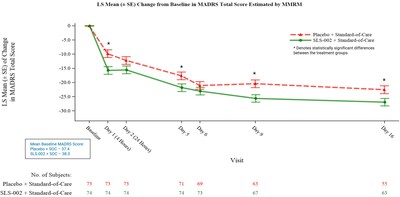
SLS-002 versus placebo demonstrated early and persistent reductions in symptoms of depression as assessed by the Montgomery-Åsberg Depression Rating Scale (MADRS)1. The graph presents results from the mixed model for repeated measures (MMRM) analysis of change from baseline in MADRS total score.
Target enrollment of this study was 220 patients, however, due to financial constraints, only 147 patients diagnosed with MDD requiring psychiatric hospitalization due to significant risk of suicide were randomized. The data from the 147 subjects (67% of target enrollment) were evaluated using the protocol-defined methods of analysis. Due to the limited sample size, the study did not meet the pre-defined primary endpoint (MADRS ANCOVA at 24 hours post dosing). However, assuming the same treatment difference and standard deviation, analyses showed that the study would have achieved statistical significance for the primary endpoint, had the study reached full enrollment (220 patients).
"The analyses of the 147 enrolled subjects in this multicenter, double-blind placebo-controlled trial of SLS-002 demonstrated both meaningful early and persistent improvement in depressive symptoms, as well as clinically meaningful reduction in acute suicidality symptoms relative to standard of care," said Tim Whitaker, M.D., Chief Medical Officer of Seelos. "We believe these results demonstrate the therapeutic potential of SLS-002 to address this huge unmet need and those at risk. We look forward to our discussions with the FDA to align on next steps. In addition, we want to thank the study participants, as well as the clinical trial sites and staff, for their expert and careful care of these high-risk study patients."
Detailed Summary of Key Efficacy Endpoints
- MADRS results at 4 hours after dosing demonstrated a statistically significant change relative to placebo (p <0.001, 5.9 point LS2 mean treatment difference)
- MADRS results at 24 hours after dosing utilizing 2-way ANCOVA3 with baseline MADRS as a covariate (the pre-defined primary endpoint/analysis) demonstrated clinically meaningful results, but did not achieve statistical significance under the methodology used (p=0.069, 3.3 point LS mean treatment difference)
- MADRS results at 24 hours after dosing utilizing an exploratory ANOVA (t-test) analysis demonstrated statistically significant change relative to placebo (p=0.049, 3.6 point mean treatment difference)
- MADRS results at Day 16 demonstrated a statistically significant change relative to placebo (p=0.012, 4.4 point LS mean treatment difference) – demonstrating persistence of effect
- Meaningful results further supported by (proportion of SLS-002 subjects versus placebo, respectively):
- MADRS Response Rate, defined as ≥ 50% reduction from baseline, at Day 16 (75.7% versus 47.9%) p<0.001
- MADRS Remission Rate, defined as a total score ≤ 12, at Day 16 (62.2% versus 32.9%) p<0.001
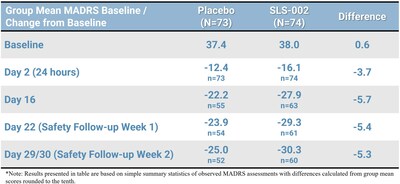
- MADRS at end of 2-week safety follow up (Day 29/30) revealed continued improvement, demonstrating no evidence of return of symptoms.
Clinically meaningful reduction in acute suicidality was demonstrated with SLS-002 over placebo. Both groups continued to improve over time.
- Sheehan-Suicidality Tracking Scale (S-STS) Total Score4: (Mean baseline total scores were 21.4 for SLS-002 and 21.0 for placebo)
- 4 hour change from baseline was -15.1 for SLS-002 and -12.0 for placebo (p=0.022)
- 24 hour change from baseline was -15.5 for SLS-002 and -12.1 for placebo (p=0.008)
- Clinical Global Impression of Severity for Suicidal Ideation and Behavior (CGIS-S/IB)5: (Mean baseline scores were 4.0 for both SLS-002 and placebo)
- 4 hour change from baseline was -1.5 for SLS-002 and -1.1 for placebo (p=0.011)
- 24 hour change from baseline was -1.7 for SLS-002 and -1.4 for placebo (p=0.102)
The table represents the observed MADRS summary statistics, including the assessments collected at the safety follow-up visits.
Detailed Summary of Safety Results
SLS-002 was well-tolerated with no new or unique safety signals identified and there were no deaths reported in the study. At least one treatment-emergent adverse event was reported in 52.7% of subjects treated with SLS-002 versus 39.7% treated with placebo; the majority of adverse events were mild or moderate and transient in nature. The most common treatment-emergent adverse events (≥5% and >placebo) were dizziness (18.9% versus 2.7%), euphoric mood (6.8% versus 0%) and suicidal ideation (5.4% versus 2.7%). There were 5 serious adverse events (3 with SLS-002, 2 with placebo), all for suicidality, all judged unrelated to the study drug, and all resolved.
Specific scales were utilized to measure the three most common known adverse events associated with ketamine treatments, which are dissociation, hemodynamic effects and sedation. The data below support that SLS-002 may reduce the frequency and severity of these most common effects compared to what is reported with other ketamine treatments.
Clinician-Administered Dissociative States Scale (CADSS)6
- At no point did the placebo-adjusted mean change from pre-dose baseline exceed the threshold of clinically meaningful dissociation (>4)
- Placebo-adjusted mean change from pre-dose baseline (at 40 minutes – correlating roughly with maximum plasma concentrations) = 3.9 after first dose and 1.8 at 1 hour post first dose
- Placebo-adjusted mean change from pre-dose baseline 2.5 on Day 4 (at 40 minutes) after second dose and 1.4 on Day 8 (at 40 minutes) after third dose
Hemodynamic Effects
- 6 subjects who received SLS-002 had an adverse event reported of either increased blood pressure or hypertension, all events were mild (except 1 moderate), all were transient and resolved
- In review of mean vital sign data for SLS-002, minimal changes were observed
- Systolic blood pressure at Baseline was 122.7 mmHg, with maximum mean values of 126.7 mmHg on days 1 and 11 (1 hour post dose)
- Diastolic blood pressure at Baseline was 77.7 mmHg, with maximum mean values of 81.1 mmHg on Day 8 (1 hour post dose)
Modified Observer's Assessment of Alertness/Sedation Scale (MOAA/S)7
- Maximum sedation (MOAA/S score < 5) occurred approximately 15 minutes after dosing on day 1 (placebo adjusted % of subjects 25.5%) with attenuation over time
"We believe these data are remarkable. We expect to move forward with our development of SLS-002 after the end of Phase II meeting with the FDA," said Raj Mehra Ph.D., Chairman and Chief Executive Officer of Seelos. "The improvements with SLS-002 were robust and continued to improve across efficacy scales with 5 doses over the two-week treatment period. In addition to the data on efficacy, the differentiated and well-tolerated safety profile of SLS-002 highly underscores this product candidate's uniqueness and potential for the treatment of ASIB in MDD. We look forward to advancing this therapy toward a potential first approval for an important unmet need for this patient population, especially considering that 2022 experienced the highest number of suicides in U.S. history."
Webcast Information
Seelos will host a webcast today, Wednesday, September 20th, at 8 a.m. ET, to discuss the results. On the call from Seelos will be Raj Mehra, Ph.D., Chairman and CEO, and Tim Whitaker, M.D., Chief Medical Officer.
Additionally, David V. Sheehan, M.D., the Distinguished University Health Professor Emeritus at the University of South Florida College of Medicine, will be on the call as well to discuss this data.
Webcast for live and replay: https://lifescievents.com/event/seelos-2
A replay of the webcast will be available shortly following the presentation.
This study randomized 147 subjects diagnosed with MDD requiring psychiatric hospitalization due to significant risk of suicide and severe depression as confirmed by the rating scales as discussed above. In addition, subjects had to have at least one suicide attempt. For more information on entry criteria visit https://clinicaltrials.gov/study/NCT04669665?term=sls-002&rank=1.
After admission to the emergency room or hospital, each subject participated in a 1- to 2‑day screening phase, a 16-day treatment phase, including clinical standard of care, during which the study drug was administered 2 times per week (total of 5 doses), and a 2-week safety follow-up phase for a total of up to 5 weeks of study participation. Subjects were treated as inpatients for approximately 7 days (including screening), and assuming the subject met readiness for discharge criteria, were discharged on Day 6 to continue the trial as outpatients. At study completion, all subjects were well-connected with follow-up care to ensure their safety.
Per the Centers for Disease Control and Prevention (CDC) provisional data, the 49,449 suicides in the U.S. in 2022 represented the highest number of suicides recorded in U.S. history and unfortunately, the medical community still lacks an FDA approved therapeutic to treat the symptoms of suicidality. According to the CDC, in 2020, suicides and non-fatal self-harm cost the U.S. over $500 billion in medical and work-loss costs, value of statistical life, and quality of life costs. Suicidal patients who present suicidal ideation and behavior symptoms at an Emergency Department and can be held in the Emergency Department for several days while awaiting an inpatient psychiatric bed. Currently in the U.S., there is a shortage of over 120,000 inpatient psychiatric beds and the average length of hospitalization for a suicidal patient is 10 days.
If you or a loved one are having thoughts of suicide, please seek immediate medical help, go to your nearest emergency room, call the Suicide and Crisis Lifeline at 988 or 1-800-273-8255 (TALK).
1 Montgomery-Åsberg Depression Rating Scale (MADRS) scale range 0-60, higher scores indicating more severe depression.
2 Least Squares (LS) Means are means estimated from a linear model that are adjusted for other effects defined in the model.
3 ANCOVA is short for Analysis of Covariance. The analysis of covariance is a combination of an ANOVA and a regression analysis.
4 Sheehan-Suicidality Tracking Scale (S-STS) is a clinician-rated scale, which includes 13 suicidality items that are rated on a scale ranging from 0 (not at all) to 4 (extremely), which yields a total score ranging from 0 to 52.
5 Clinical Global Impression of Severity for Suicidal Ideation and Behavior (CGIS-SI/B) is a 5-point clinician-rated measure of suicidality-specific symptom severity, ranging from 1 (not at all suicidal) to 5 (among the most extremely suicidal).
6 Clinician-Administered Dissociative States Scale (CADSS) is a standardized instrument used to measure present-state dissociative symptoms. The scale includes 23 subjective items to be answered by the subject according to a 5-point scale (0 = not at all, 1 = mild, 2 = moderate, 3 = severe, and 4 = extreme). CADSS total score range is 0-92; a higher score reflects a more severe condition.
7 Modified Observer's Assessment of Alertness/Sedation Scale (MOAA/S) is a 6 point scale with a score of 5 defined as responds readily to name, and a score of 0 defined as does not respond to painful stimulus.
About SLS-002
SLS-002 is intranasal racemic ketamine with two investigational new drug applications for the treatment of Acute Suicidal Ideation and Behavior in Major Depressive Disorder and in Post-Traumatic Stress Disorder. SLS-002 was originally derived from a Javelin Pharmaceuticals, Inc./Hospira, Inc. program with 16 clinical studies involving approximately 500 subjects. Seelos looks to address an unmet need for a therapy to treat suicidality in the U.S. with SLS-002. Traditionally, anti-depressants have been used in this setting but many of the existing treatments are known to contribute to an increased risk of suicidal thoughts in some circumstances, and if they are effective, it often takes weeks for the full therapeutic effect to be manifested. Based on information gathered from the databases of the Agency for Healthcare Research and Quality, there were more than 1,000,000 visits to emergency rooms for suicide attempts in 2019 in the U.S. alone. Experimental studies suggest ketamine has the potential to be a rapid, effective treatment for refractory depression and suicidality.
About Seelos Therapeutics
Seelos Therapeutics, Inc. is a clinical-stage biopharmaceutical company focused on the development and advancement of novel therapeutics to address unmet medical needs for the benefit of patients with central nervous system (CNS) disorders and other rare diseases. The Company's robust portfolio includes several late-stage clinical assets targeting indications including Acute Suicidal Ideation and Behavior (ASIB) in Major Depressive Disorder (MDD), amyotrophic lateral sclerosis (ALS) and spinocerebellar ataxia (SCA), as well as early-stage programs in Huntington's disease, Alzheimer's disease, and Parkinson's disease.
For more information, please visit our website: http://seelostherapeutics.com, the content of which is not incorporated herein by reference.
Forward Looking Statements
Statements made in this press release, which are not historical in nature, constitute forward-looking statements for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. These statements include, among others, those regarding Part 2 of the Phase II study of SLS-002, statements regarding SLS-002's prospects and potential, statements regarding any potential market opportunity for SLS-002, statements to the effect that Part 2 of the Phase II study of SLS-002 would have achieved statistical significance for the primary endpoint had the study reached full enrollment and statements regarding Seelos' development plans for SLS-002 and any planned meetings and discussions with the FDA. These statements are based on Seelos' current expectations and beliefs and are subject to a number of risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Risks associated with Seelos' business and plans described herein include, but are not limited to, the risk of not successfully executing its preclinical and clinical studies, not being able to move forward with the development of SLS-002 after the anticipated end of Phase II meeting with the FDA, and not gaining marketing approvals for SLS-002 and/or its other product candidates; the risk that prior clinical results may not be replicated in future studies and trials (including the risk that the results from the prior studies of SLS-002 may not be replicated or may be materially different from the results of Part 2 of the Phase II study of SLS-002); the risks that clinical study results may not meet any or all endpoints of a clinical study and that any data generated from such studies may not support a regulatory submission or approval; the risks associated with the implementation of Seelos' business strategy; the risks related to raising capital to fund its development plans and ongoing operations; the risks related to Seelos' current stock price; as well as other factors expressed in Seelos' periodic filings with the U.S. Securities and Exchange Commission, including its most recent Annual Report on Form 10-K and Quarterly Reports on Form 10-Q. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, even if subsequently made available by us on our website or otherwise. We do not undertake any obligation to update, amend or clarify these forward-looking statements, whether as a result of new information, future events or otherwise, except as may be required under applicable securities laws.
Contact Information:
Anthony Marciano
Chief Communications Officer
Seelos Therapeutics, Inc. (Nasdaq: SEEL)
300 Park Avenue
New York, NY 10022
(646) 293-2136
anthony.marciano@seelostx.com
https://seelostherapeutics.com/
https://twitter.com/seelostx
https://www.linkedin.com/company/seelos
Mike Moyer Managing Director
LifeSci Advisors, LLC
250 West 55th St., Suite 3401
New York, NY 10019
(617) 308-4306
mmoyer@lifesciadvisors.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/seelos-therapeutics-announces-top-line-results-from-sls-002-phase-ii-study-in-adults-with-major-depressive-disorder-at-imminent-risk-of-suicide-301932877.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/seelos-therapeutics-announces-top-line-results-from-sls-002-phase-ii-study-in-adults-with-major-depressive-disorder-at-imminent-risk-of-suicide-301932877.html
SOURCE Seelos Therapeutics, Inc.